The SPICA Technology
The SPICA Technology
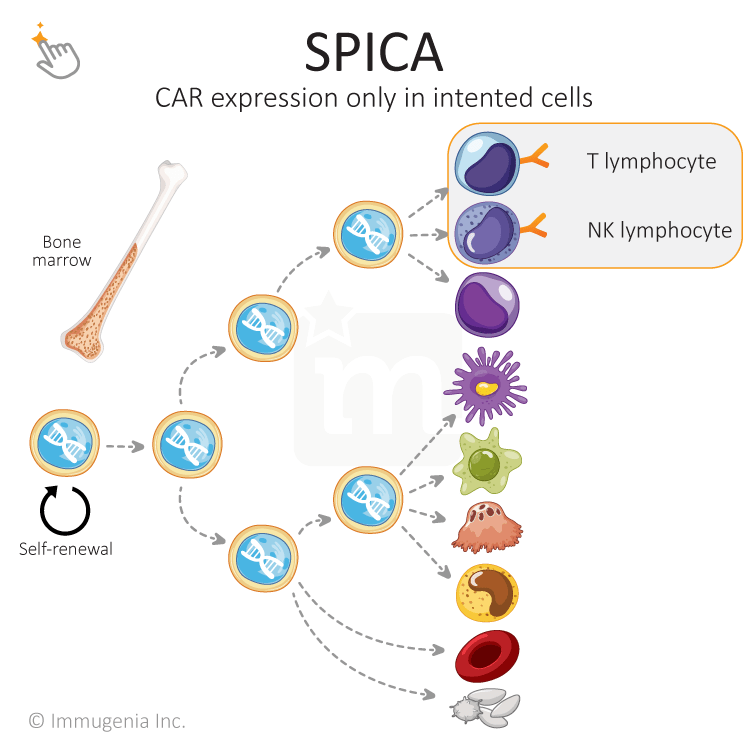
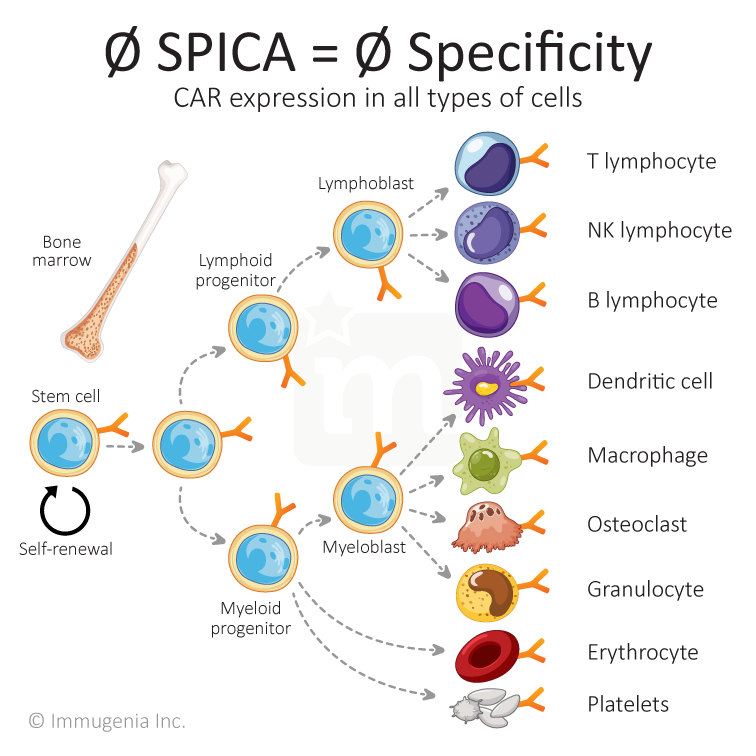
At Immugenia, we have created a biotechnological tool enabling the use of hematopoietic stem cells to treat life-threatening cancers. Our SPICAs (Specific Promoters for Immune Cell Activity) are a variety of synthetic promoters that drives the expression of a gene only in intended cells, thereby adding selectivity and allowing the use of stem cells for therapy.
A promoter is a sequence that turns a gene on/off. Those promoters dictate which proteins are expressed in a given cell, allowing for a cell to be specialized in its function. When a cell is genetically modified with a CAR or another added gene, the new gene is expressed in all types of cells because the used promoter is not tailored to be selective. This precludes the use of stem cells for immunotherapy. Immugenia has solved this technological roadblock and unlocked the power of stem cells.

At Immugenia, we have created a biotechnological tool enabling the use of hematopoietic stem cells to treat life-threatening cancers. Our SPICAs (Specific Promoters for Immune Cell Activity) are a variety of synthetic promoters that drives the expression of a gene only in intended cells, thereby adding selectivity and allowing the use of stem cells for therapy.
A promoter is a sequence that turns a gene on/off. Those promoters dictate which proteins are expressed in a given cell, allowing for a cell to be specialized in its function. When a cell is genetically modified with a CAR or another added gene, the new gene is expressed in all types of cells because the used promoters is not tailored to be selective. This precludes the use of stem cells for immunotherapy. Immugenia has solved this technological roadblock and unlocked the power of stem cells.
Therapeutic Strategy
One immunotherapy for life
Therapeutic Strategy
One immunotherapy for life
Chimeric Antigen Receptors (CAR) are molecule tailored to recognize tumor cells and trigger its death.
Traditionally CAR are put into T cells as these white blood cells have a capacity to kill other cells. Impressive results have been obtained with patients suffering from leukemias and lymphomas, but the technology is still in its infancy and needs to be improved to face its numerous current limitations.
At Immugenia we are creating CAR therapies based on the modification of hematopoietic stem cells, the cells that give rise to all the blood cells, white (immune) and red. Everyone, even patients going through aggressive regiment of chemotherapy, have hematopoietic stem cells that are easily harvestable to be modified.
Once transplanted back in their body, patients will produce their own CAR T and NK cells. Since stem cells have the ability to self-renew and to give rise to progeny for life, this therapy would be “One immunotherapy for a lifetime!”
Chimeric Antigen Receptors (CAR) are molecules tailored to recognize tumor cells and trigger their death.
Traditionally CAR are put into T cells as these white blood cells have a capacity to kill other cells. Impressive results have been obtained with patients suffering from leukemias and lymphomas, but the technology still faces some challenges reducing its efficacy in the long term and causing side effects.
At Immugenia we are creating CAR therapies based on the modification of hematopoietic stem cells, the cells that give rise to all the blood cells, white (immune) and red. Everyone, even patients going through aggressive regiment of chemotherapy, have hematopoietic stem cells that are easily harvestable to be modified.
Once HSCs have been engineered, patients will produce their own CAR T and NK cells. Since stem cells have the ability to self-renew and to give rise to progeny for life, this therapy would be “One immunotherapy for a lifetime!”
Current challenges in CAR T-cell therapy
Current challenges in CAR T-cell therapy
Advantages of HSC-based immunotherapies
Advantages of HSC-based immunotherapies
In vivo CAR delivery
The future of CAR therapy
In vivo CAR Delivery
The future of CAR therapy
While CAR-T therapy has demonstrated effectiveness, its widespread adoption is constrained by high costs, the need for personalized treatment, and the complexities of ex vivo cell modification. A promising approach to improve accessibility is to replace the ex vivo modification and expansion process with an in vivo method. This involves systemically administering a virus that transduces T, NK or HSC to express CARs directly within the patient’s body.
Various gene therapy delivery vehicles have been developed for this purpose. However, to date, only a few have achieved adequate specificity in targeting the desired cells, posing a risk of unintended genetic modification in other organs, such as the liver.
At Immugenia, our advanced synthetic promoter technology allows for targeted expression of the transgene in specific cell types. This innovation could be the crucial element needed to make in vivo CAR delivery feasible. By combining these synthetic promoters with lentiviral-type delivery systems targeting immune cells, we aim to enhance the specificity of CAR expression, even if the delivery system inadvertently enters the wrong cells (off-target), thereby enabling safer in vivo CAR delivery.
No need for a perfect drug delivery system
for specific gene expression
Benefits
While CAR-T therapy has demonstrated effectiveness, its widespread adoption is constrained by high costs, the need for personalized treatment, and the complexities of ex vivo cell modification. A promising approach to improve accessibility is to replace the ex vivo modification and expansion process with an in vivo method. This involves systemically administering a virus that transduces T, NK or HSC to express CARs directly within the patient’s body.
Various gene therapy delivery vehicles have been developed for this purpose. However, to date, only a few have achieved adequate specificity in targeting the desired cells, posing a risk of unintended genetic modification in other organs, such as the liver.
At Immugenia, our advanced synthetic promoter technology allows for targeted expression of the transgene in specific cell types. This innovation could be the crucial element needed to make in vivo CAR delivery feasible. By combining these synthetic promoters with lentiviral-type delivery systems targeting immune cells, we aim to enhance the specificity of CAR expression, even if the delivery system inadvertently enters the wrong cells (off-target), thereby enabling safer in vivo CAR delivery.